Why Must the Mass of an Anhydrous Salt Be
Mass of substances before reaction Mass of substances after reaction In this experiment. Molar mass of MgSO4 anhydrous Molar mass of MgSO4 anhydrous is 1203676 gmol.

Use The Information Given On The Nickle Ii Sulfate To Do The Calculations Calculations 1 Calculate The Formula Homeworklib
Using the gravimetric analysis method the sample salt was measured heated and was measured again after cooled down to room temperature.

. Anhydrous means without water. 1 Inorganic salt hydrates as PCMs. See answer 1 Best Answer.
Use a chemistry source to find the number of water molecules bound per formula unit then write the correct formula for hydrated salt of the following. The moles of salt should be the smaller of the two values. Anhydrous Compounds Anhydrous chemicals do not contain water and are typically stored in a container with a desiccator.
Because once it starts cooling it begins collecting water again. This is called heating to constant mass. Anhydrous substances exist in solid liquid and gas forms.
To prevent breakage when cold tongs touch hot crucibles. 1Determine the mass of the hydrate used. If we heat hydrated salts they emit water molecules as water vapor.
Hydrate Heat ---. Table salt is anhydrous sodium chloride NaCl. The sample must be heated at least two times to assure that ail of the water has been removed from the hydrated salt.
Mass of dish and hydrate - mass of dish and anhydrous salt 503 g-495 g 080 g. Hydrated salt contains water molecules chemically bound to it while anhydrous salt is a substance that fire can readily remove the water molecules. Why must dish be cooled before finding mass.
The total mass remains constant. In the experiment the percentage of water in Copper ll Sulfate was the purpose of the lab. Track your food intake exercise sleep and meditation for free.
Heating copper II sulfate pentahydrate CuSO 4 5H 2 O yields anhydrous copper II sulfate CuSO 4. Because once it starts cooling it begins collecting water again. Hydrated salts or hydrates contain a certain amount of water within their molecular structures.
Mass of Hydrate mass of H. Why must the mass of the anhydrous salt be measured immediately upon cooling. Nickel II nitrate d.
Chemistry Measurement SI System of Measurement. Once cool though it can start to collect water again from the atmosphere and gain mass. To find this the mass of the hydrate and mass of the water lost was calculated.
If hydrate was not heated long enough percent of water value would be. To find the number of moles of anhydrous salt remaining in the crucible I took the mass of anhydrous salt 0908 g and divided that by its molar mass 110. Divide the moles of water by the moles of salt to find the ratio.
Hydrate Anhydrous Salt Water The percent of water in a hydrate can be found experimentally by accurately determining the mass of the hydrate and the mass of the anhydrous salt. 2010-10-22 19. Water in the air could loosely bond with the salt remaining throwing off the true mass.
Why must the mass of the anhydrous salt be measured somewhat quickly after cooling. This will allow you to find the mole ratio. This is why weighing a anahydrous salt at just the right time is essential.
Why must the mass of an anhydrous salt be measured immediately upon cooling. The difference in mass is due to the water lost by the hydrate. Get control of 2021.
The key difference between hydrated salt and anhydrous salt is that the hydrated salt molecules are attached to water molecules whereas the anhydrous salt molecules are not attached to any water molecules. 2Determine the mass of the water lost. The mass of the crucible was 17985 g.
The law of definite proportions or constant composition states that the elements in a pure compound are present in a definite constant. Common ratios will range from 12 up to 110. Mass of dish and anhydrous salt 495 g.
This procedure allowed the weight of water lost in the salt to be obtained. If you measure the mass of the salt while hot or warm the mass will constantly change because its giving off something like water. How do you calculate the mass of the hydrate heated.
M 2009 MM 12 493712 gimol. 1 Answer anor277 Nov 9 2016 21447-17985gg. Mass of hydrate - mass of water lost.
B Why must the crucible containing the hydrated salt not be heated to redness. Heating to constant mass ensures that all of the water of hydration has been driven off and we are massing only the anhydrous salt. This water is called the water of hydration and without it a salt is anhydrous.
To eliminate the effects of warm air so that you can get an accurate mass measure In the water in a hydrate experiment why must the mass of the anhydrous salt be measured immediately upon cooling. When you apply heat to these salts water is released. The mass after the second heating should be within 005 g of the first heating.
Salts are compounds containing anions and cations in a crystalline form. Why must anhydrous salt be measured immediately upon cooling. Convert between MgSO4 anhydrous weight and moles.
2- A- If 2000g of CuSO45H2O is dehydrated what is the mass of the anhydrous salt remaining. O lost mass of anhydrous salt after heating. The question asked you to determine the mass of the hydrate.
Because salt tends to naturally bind to moisture anhydrous salt usually has to be processed. Why must you measure the mass of the anhydrous salt immediately after it is cool. Convert the mass of the anhydrous salt m2 by dividing it by the molar mass of the anhydrous salt.
The general reaction for heating a hydrate is. In the water in a hydrate experiment why must the mass of the anhydrous salt be measured immediately upon cooling. Mass of dish and hydrate - Mass of dish 503 g-483 g200g.
Why must the mass of an anhydrous salt be. Divide the mass of water lost when you heated the salt by the molar mass of water roughly 18 grams per mole. The sample must be heated twice so that the difference between the two masses can be used to determine how much salt is in the sample.
The mass of the crucible and the anhydrous salt was 20070 g. What is a hydrate. Gaseous HCl is anhydrous which differentiates it from hydrochloric acid a solution of 37 percent HCl in water ww.
Rearranges the combination of atoms.

Solved Lass Of Anhydrous Salt Moles Of Anhydrous Salt Mass Chegg Com

Solved I Cant Figure Out The Mass Of Anhydrous Salt That Chegg Com

Ap Chemistry Question 504 Answer And Explanation Crackap Com

Answered The Hydrated Salt My3 Xh2o Was Heated Bartleby

Solved Can You Help Fill In Some Of The Blanks With The Data Chegg Com

Solved Mass Of Anhydrous Salt 7 2 Moles Of Anhydrous Salt Chegg Com

Solved Calculate The Mass Of Anhydrous Salt After Final Chegg Com

Solved Mass Of Anhydrous Salt 7 2 Moles Of Anhydrous Salt Chegg Com
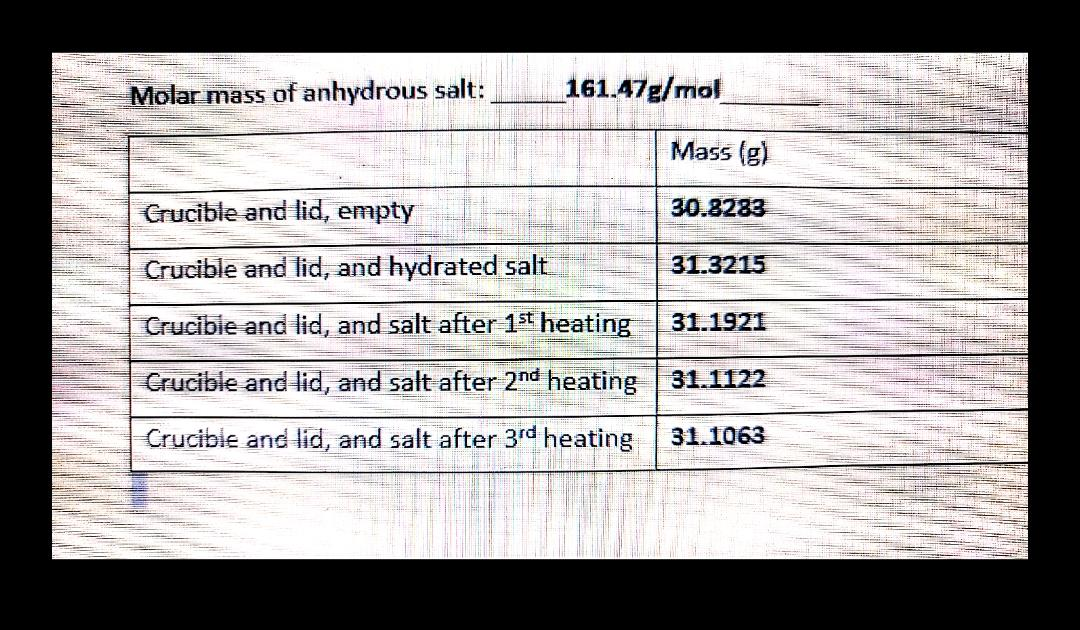
Solved A Calculate The Mass Of The Anhydrous Chegg Com

Unknown Number Data Trial 1 Trial 2 Mass Of The Crucible And Cover 28 2569 Mass Of Homeworklib

Use The Information Given To Calculate The Formula For Mass Molar Mass Of The Anhydrous Salt Of Homeworklib

Calculations 1 Calculate The Formula Mass Molar Chegg Com

Solved 4 Mass Of Crucible And Anhydrous Salt After The Chegg Com

Solved Calculate The Mass And Moles Of The Anhydrous Salt Chegg Com

Solved 10 Add A Few Drops Of Water To The Anhydrous Salt Chegg Com

Formula Of Anhydrous Salt Used Formula Of The Salt Chegg Com

Solved Molar Mass Of Anhydrous Salt Empty Mass Mass With Chegg Com


Comments
Post a Comment